|
One lesson, learned the hard way, was the difficulty in keeping track of progress when there was one big 20-page checklist. Most facilities create a checklist of tasks for which new staff members will receive training. 2 That brochure outlines the requirements for competency assessment, including those individuals who may assess the competency of others. 
In December 2012 CMS published CLIA Brochure 10, What Do I Need to Do to Assess Personnel Competency? in order to improve competency assessments in the field. Documentation is the way to prove that staff is trained. 1 Both regulations in the good manufacturing practices section of the Code of Federal Regulations (21 CFR 211 and 21 CFR 600-640) and the regulations implementing the Clinical Laboratory Improvement Act (CLIA) in 42 CFR 493 require trained staff. In addition, staff must receive training on the current good manufacturing practices regulations that apply to their role. 
List and explain which surgical procedures would require T/S or T/C to be performed preoperatively.įDA regulations require that staff be trained to perform their duties.Understand the importance of MSBOS implementation in an institution.Define the terminology associated with MSBOS.Understand what procedural drift is and describe different ways to monitor it in the laboratory.Understand the importance of efficiently assessing laboratory personnel competency during the first year of employment and beyond.Upon completion of this article, the reader will be able to: The suggestions below represent the lessons learned from experiences in assessing employee competency. The competency assessment tools described are most relevant for moderately complex and highly complex tests. This has been termed “procedural drift.” This article will review how we have used competency assessments and other audits in our transfusion service at the University of Michigan Hospitals and Health Centers to verify that staff members are competent and performing their duties according to facility-defined procedures. 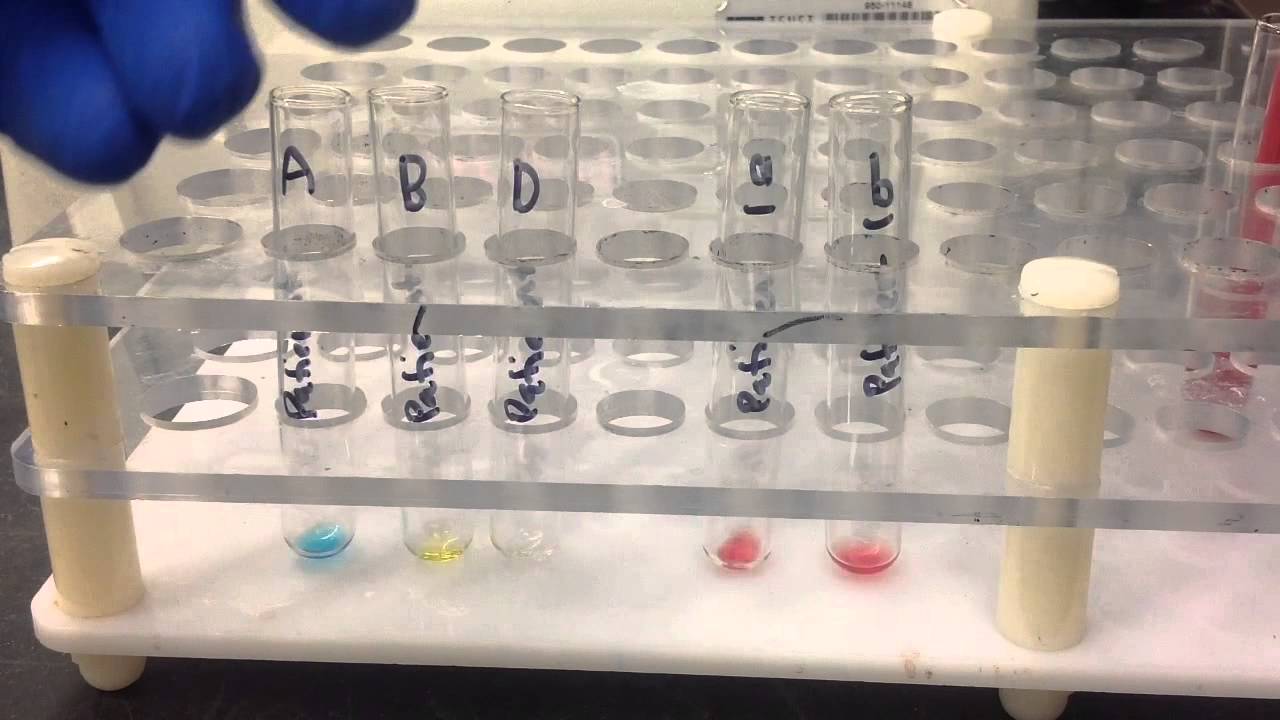
Over time, staff members may have introduced personal modifications to tasks in order to improve productivity or reduce costs. Management may be responsible for ensuring that immunohematology procedures are consistent with equipment and that reagent manufacturers’ instructions for use are in compliance with regulatory agency requirements and meet the individual facility’s requirements, but it is the transfusion service technologists who are critical: they are actually performing the tests. Compliance with regulatory and accrediting agency requirements is dependent on the competency and willingness of transfusion service staff members to perform their duties as instructed.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
